When we think of the placenta (afterbirth), it is easy to imagine a simple connection. A one-way transport system of nutrients from mother to baby. Our focus is often drawn to the developing baby, but lately there has been growing interest in the placenta and the cells that orchestrate its function. After all, the placenta can survive independently, but the growing baby cannot develop in the womb without the placenta. Errors in placental formation can lead to miscarriage or growth restriction in babies. Placental dysfunction can even have effects into adulthood with increased rates of cardiovascular disease. This blog post will introduce the functions of the placenta, some of the cells involved, and the impact of placental development on long-term health.
Initiation of placental development
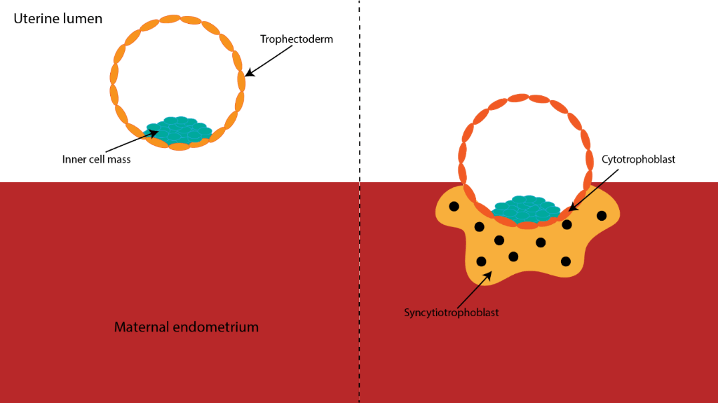
The journey starts as the embryo begins to invade into the mother’s uterus (endometrium), this occurs around a week after fertilisation. The embryo is broadly defined into two parts at this stage: the inner cell mass which will form the baby, and the trophectoderm which will form the placenta.
As the embryo implants in the endometrium and begins to invade, the trophectoderm forms two key cell types: cytotrophoblasts and syncytiotrophoblasts (Figure 1). Some of the cytotrophoblasts fuse to form syncytiotrophoblasts. Syncytiotrophoblasts contain many nuclei and invade into the endometrium. Just below the surface of the implantation site, glands and spiral arteries are waiting to provide the growing embryo with nutrition throughout pregnancy.1 During pregnancy, the endometrium of the mother is transformed into the decidua, this is an adaptation of the uterus to allow adequate nutrition of the fetus by increasing gland secretions, glycogen storage in cells, and recruitment of immune cells.
Establishing nutrition from the mother to the placenta
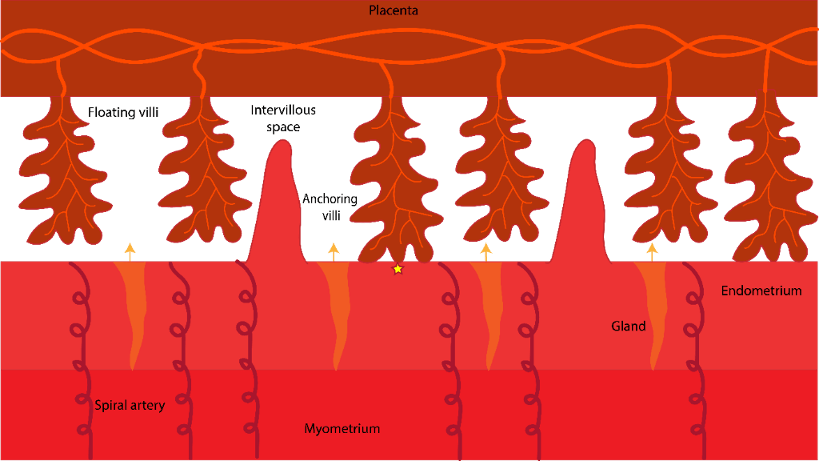
The placenta is a ‘gatekeeper’, controlling what is transported to the fetus and what is transferred to the mother. The placenta forms branched structures called villi. The villi contain blood vessels which transport nutrients and gases to the developing fetus. Waste products including carbon dioxide and urea are transferred to the mother. Hormones are also secreted by the placenta into the maternal blood stream which impact lactation (prolactin), labour (relaxin), and even maternal bonding with the baby (oxytocin).2 Figure 2 shows the placenta and the maternal endometrium during the first trimester; placental villi stretch out into pools of maternal gland secretions. The space between the placental villi is known as the intervillous space. Villi in contact with the endometrium are known as anchoring villi.
In the first trimester, spiral arteries are ‘plugged’ by placental cells to prevent blood flowing into the intervillous spaces. Instead, gland secretions provide nutrition (yellow arrows in Fig.2). This is a clever solution, as developing organs in the fetus require a low oxygen environment during the first trimester. This also allows time to develop effective protection against harmful free radicals which are generated when cells use oxygen. In the second and the third trimesters, maternal blood from uterine spiral arteries enter the intervillous space.
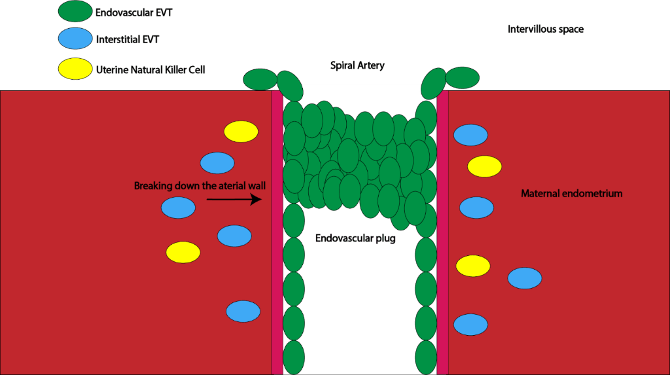
The process by which placental cells alter spiral artery structure and function is termed ‘spiral artery remodelling’ (Figure 3).3 Extra-villous trophoblast cells (EVTs) invade the maternal endometrium from the anchoring villi of the placenta. There are two key types of EVT involved in spiral artery remodelling: endovascular EVTs and interstitial EVTs. Endovascular EVTs invade into the spiral artery, replace the arterial epithelium, and form an endovascular plug. The plug prevents red blood cells from entering the extra-villous spaces. While the vessels are plugged, interstitial EVTs and uterine natural killer cells break down smooth muscle cells and elastic filaments of the vessels. This allows for a low-pressure, high-volume flow of maternal blood into the intervillous space once the endovascular plug disintegrates at the beginning of the second trimester.
What happens when this goes wrong?
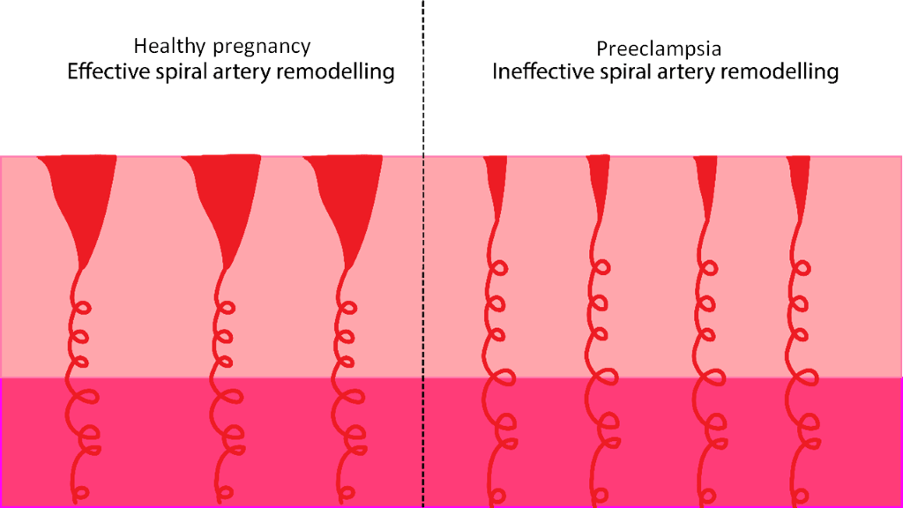
2-8% of pregnant women experience preeclampsia.4 Incomplete spiral artery remodelling is widely thought to contribute to preeclampsia (Figure 4). In incomplete spiral artery remodelling the arterial walls have not been broken down effectively. Therefore, the arterial muscles are still able to contract and divert blood away from the placenta. This leads to fluctuations in the oxygen transferred to the placenta and subsequently to the fetus. A rise in blood pressure, excess protein in urine (proteinuria), and tissue swelling with water (oedema) is observed in the mother. Around 20% of women with preeclampsia have low birth weight babies. If preeclampsia is left untreated it can develop into eclampsia which manifests in seizures and can be fatal to both mother and baby.
Fetal growth restriction (FGR), caused by placental insufficiency in conditions such as preeclampsia, increases the risk of developing cardiometabolic disorders like diabetes and cardiovascular disease in adulthood. Additionally, FGR increases the risk of neurocognitive disabilities such as cerebral palsy.5 FGR affects around 10% of pregnancies worldwide, therefore it is an important area of research to improve population health.
Despite the importance of preeclampsia and FGR in maternal and fetal health, we still do not know conclusively what happens at the cellular level. My research focuses on investigating the effects of cellular stress on the proportions of trophoblast cell types that develop during implantation. By studying placental development, we can shed light on the complex relationships which contribute to conditions including preeclampsia. Therefore, placental research has far-reaching implications for both maternal and fetal health and the population at large.
References
1. Aplin JD. Implantation. In: Encyclopedia of Hormones. Elsevier; 2003:289-297.
2. Gude NM, Roberts CT, Kalionis B, King RG. Growth and function of the normal human placenta. Thromb Res. 2004;114(5-6 SPEC. ISS.):397-407. doi:10.1016/j.thromres.2004.06.038
3. Sato Y. Endovascular trophoblast and spiral artery remodeling. Mol Cell Endocrinol. 2020;503:110699. doi:10.1016/j.mce.2019.110699
4. Beaufils M. Pregnancy hypertension. Nephrol Ther. 2010;6(3):200-214. doi:10.1016/j.nephro.2010.03.005
5. Colella M, Frérot A, Novais ARB, Baud O. Neonatal and Long-Term Consequences of Fetal Growth Restriction. Curr Pediatr Rev. 2018;14(4):212-218. doi:10.2174/1573396314666180712114531
Discover more from Research Hive
Subscribe to get the latest posts sent to your email.
